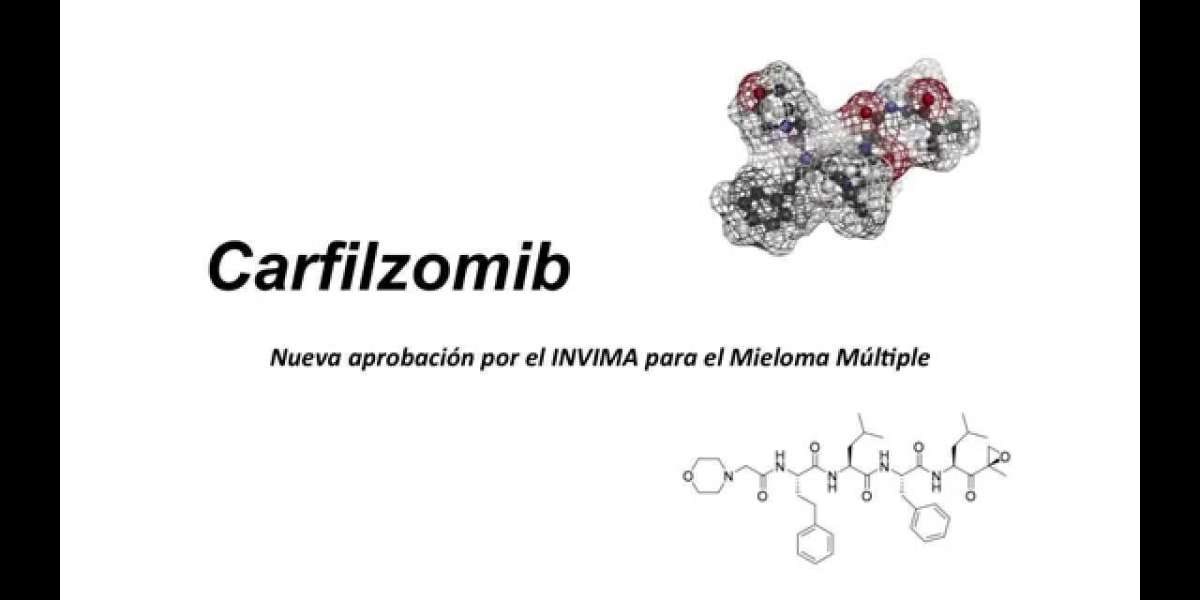
Carfilzomib Market Defination:
The Carfilzomib Market refers to the economic and clinical landscape surrounding the pharmaceutical drug carfilzomib. Carfilzomib is a proteasome inhibitor used primarily in the treatment of multiple myeloma, a type of cancer affecting plasma cells in bone marrow. It functions by selectively inhibiting the proteasome, a complex protein-degrading machinery essential for cell function and survival. This inhibition leads to the accumulation of proteins within cancer cells, triggering cell death through apoptosis.
Print This Guide for Future Reference: https://wemarketresearch.com/reports/request-free-sample-pdf/carfilzomib-market/1493
Exploring the Carfilzomib Market: Advancements in Multiple Myeloma Treatment
In the realm of oncology, particularly in the treatment landscape of multiple myeloma, carfilzomib has emerged as a cornerstone therapy, offering new hope and improved outcomes for patients. This blog delves into the dynamic carfilzomib market, examining its impact, current trends, challenges, and future prospects.
Understanding Carfilzomib
Carfilzomib is a proteasome inhibitor approved for the treatment of relapsed or refractory multiple myeloma. It works by selectively and irreversibly binding to the 20S proteasome, disrupting protein degradation in cancer cells and inducing apoptosis. Approved by the FDA in 2012, carfilzomib has since been integrated into treatment protocols, often in combination with other agents like lenalidomide and dexamethasone.
Market Dynamics
- Current Landscape: The Carfilzomib Market is driven by its efficacy in treating relapsed or refractory multiple myeloma, particularly in patients who have received prior therapies. Its mechanism of action and clinical benefits have positioned it as a valuable option in the treatment algorithm for multiple myeloma.
- Treatment Advancements: Clinical studies have demonstrated that carfilzomib-based regimens prolong progression-free survival and overall survival compared to traditional therapies. Its approval marked a significant advancement in the management of multiple myeloma, offering a targeted approach to combating the disease.
- Competitive Environment: Within the proteasome inhibitor class, carfilzomib competes with bortezomib and ixazomib, each offering unique profiles in terms of efficacy, safety, and administration convenience. Ongoing research aims to optimize carfilzomib’s use through novel combinations and sequencing strategies to maximize patient benefit.
Clinical Applications
- Approved Indications: Carfilzomib is primarily indicated for use in combination with other agents for the treatment of relapsed or refractory multiple myeloma. Clinical trials are also exploring its potential in newly diagnosed patients and maintenance therapy settings, broadening its scope of application.
- Future Directions: Research efforts are focused on expanding carfilzomib’s indications and understanding its synergies with emerging therapies such as immunomodulators, monoclonal antibodies, and cellular therapies like CAR-T cells. These endeavors aim to further improve treatment outcomes and offer personalized therapeutic approaches.
Carfilzomib Market Challenges and Opportunities
- Challenges: Economic considerations remain a significant challenge in the adoption of carfilzomib, given its high cost as a biologic therapy. Managing treatment-related adverse events, such as cardiovascular complications and hematologic toxicities, also requires vigilant monitoring and proactive management strategies.
- Opportunities: Advances in biomarker identification and personalized medicine offer opportunities to tailor carfilzomib-based therapies to individual patient profiles. Moreover, ongoing research into combination therapies and novel formulations aims to enhance efficacy while minimizing adverse effects, thereby improving patient adherence and outcomes.
Patient Impact and Healthcare Considerations
- Patient Experience: For patients diagnosed with relapsed or refractory multiple myeloma, carfilzomib represents a crucial treatment option that can potentially extend survival and improve quality of life. Education and support programs play a vital role in helping patients manage treatment-related challenges and adhere to therapy.
- Healthcare System Implications: Integrating carfilzomib into clinical practice requires healthcare providers to navigate complex treatment algorithms and ensure appropriate patient monitoring. Collaboration among multidisciplinary teams, including oncologists, hematologists, and supportive care specialists, is essential for optimizing patient care and outcomes.
Regulatory and Market Access
- Regulatory Landscape: Regulatory approvals and reimbursement policies influence the accessibility of c Carfilzomib Market in different regions. Streamlining regulatory processes and demonstrating cost-effectiveness through real-world evidence are crucial for enhancing market access and patient affordability.
- Market Expansion: As clinical data continues to evolve and new indications are explored, the carfilzomib market is poised for growth. Market expansion strategies should prioritize evidence-based medicine and stakeholder collaboration to drive adoption and improve patient access.
Conclusion
In conclusion, the carfilzomib market represents a significant advancement in the treatment of multiple myeloma, reflecting the transformative impact of targeted therapies in oncology. Its approval and integration into treatment protocols underscore a shift towards personalized medicine and multidisciplinary care approaches that optimize patient outcomes.
While challenges such as economic considerations and treatment-related adverse events persist, ongoing research and collaborative efforts among stakeholders are paving the way for continued innovation and improvement in Carfilzomib-Based Therapies. By addressing these challenges proactively, healthcare providers and pharmaceutical companies can ensure that carfilzomib realizes its full potential in improving the lives of patients battling multiple myeloma.
Stay informed and engaged with the latest developments in the carfilzomib market to contribute to advancements in oncology and patient-centered care.